Everything you need to know about stem cells
What are stem cells, and what makes them so unique? What are the different types of stem cells, and how have they impacted modern medical science? Here is us answering all your questions about stem cells.

Imagine holding a burning matchstick in your hand. You can sense the sharp edges of the small wooden stick at the tip of your fingers. However, your fingers are more occupied with the bright flame inching closer. Soon you begin to feel the painful, burning sensation of the flame, and you quickly drop the matchstick down lest you burn your fingers!
Relatable? - Two main things happened here: The pressure exerted by the matchstick on your skin induced a sensation of touch, and the flame induced a sensation of burning pain.
Now broadly speaking, we know how this worked. The wood and the flame were sensed by the nerves in your fingers, which sent hurrying signals to your brain, which in turn responded - ‘Drop the matchstick immediately!’. Your fingers were consequently saved from a burn injury. But how did these minute nerve cells, called neurons, differentially detect the surface of the wooden matchstick and the heat of the flame?
This year’s Nobel laureates in physiology and medicine, David Julius and Ardem Patapoutian, unravel unique ‘sensors’ present in our neurons that can detect pressure and heat.
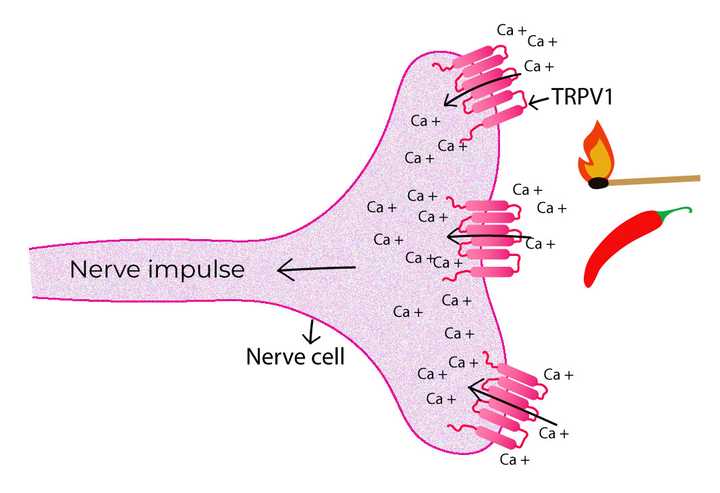
The protein receptor TRPV1 senses heat and capsaicin by allowing the influx of calcium ions. The influx results in an electrochemical current which triggers a nerve impulse.© Sunaina Rao.
Sensory information in our environment, which could be in the form of light, sound, taste, smell and touch, is essentially detected by sensory neurons present in our sense organs. This information, called a stimulus, is transmitted to the central nervous system (CNS), consisting of the spinal cord and brain. Here the stimulus is processed, and appropriate responses are transmitted back to the muscles via motor neurons. The motor neurons in turn bring about appropriate muscle movement (for example, the muscle movement that caused you to drop the matchstick).
Interestingly, there exists a special category of sensory neurons that are assigned to sensing pain. These are called nociceptors. What do nociceptors do? - They sense specific stimuli which are ‘noxious’ or harmful. Examples of this can be a harmful chemical, or the prick of a pin, or a burning flame like the one we saw before.
Nociceptors have in fact been studied for a long time now and are described by their sensitivity to a certain chemical called capsaicin. Now knowingly or unknowingly, we all are familiar with capsaicin. Capsaicin is a highly pungent substance present in chilli peppers responsible for causing that burning sensation when we bite into a chilli. Several biochemical studies have tried to understand how nociceptors sense capsaicin, but until two decades ago we had no idea.
David Julius and his team in the late 1990s started investigating this unique interaction between nociceptors and capsaicin. They guessed that these neurons must interact with capsaicin via some kind of protein present in the outer membrane of the neuron. To test this, they isolated over 16,000 candidate genes from the nociceptors that might code for this protein. They then introduced them into non-neural cells (cells that are not nerves) which showed no sensitivity to capsaicin. The right gene must be able to render the non-neural cells sensitive to capsaicin - A simple yet extremely tedious experiment!
Fortunately, their meticulous work paid off when they identified a specific gene clone that was actually able to confer capsaicin sensitivity in the non-neural cells. They discovered that this gene indeed coded for a protein on the cell membrane. On exposure to capsaicin, this channel-like receptor protein opened up and allowed the entry of calcium ions. This inflow created an electrochemical current in the neurons, thereby sensitising them to capsaicin.
What was even more interesting was that not only was this protein responsive to capsaicin, but it was also sensitive to harmful temperatures above 43℃. Mice that were designed to lack this protein in their neurons could not detect capsaicin or high temperatures!
This remarkable protein was later named TRPV1.
The cooling effect of menthol present in countless mints is likely familiar to all of us. If our neurons contain specific proteins that sense heat, we can safely guess that they might also contain distinct proteins that sense the cooling effects of certain compounds like menthol, and cold temperatures in general.
Studies conducted by David Julius and Ardem Patapoutian and their respective teams independently uncovered yet another unique protein in neurons. This one was receptive to menthol and cold temperatures - between 8℃ and 28℃. Just like TRPV1, this protein, which they named TRPM8, was a channel protein on the cell membrane that allowed an influx of calcium ions on exposure to cooling temperatures.
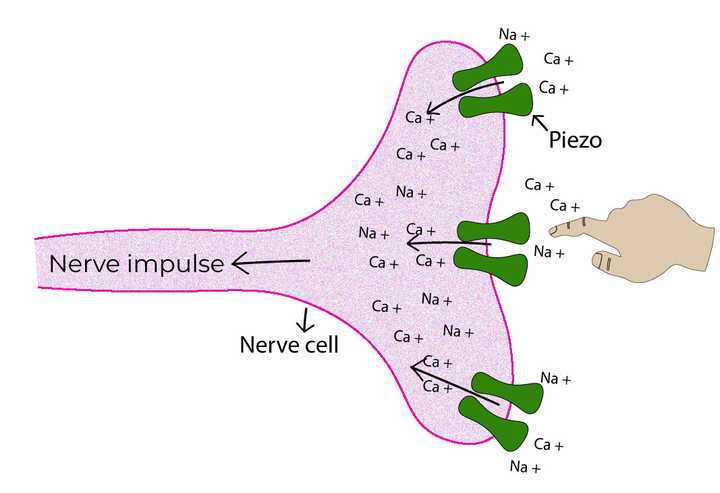
The protein receptor Piezo senses pressure by allowing the influx of calcium and sodium ions. The influx results in an electrochemical current which triggers a nerve impulse. © Sunaina Rao.
So far we have seen how specific proteins in neurons help sense temperature. But how do our nerves sense mechanical pressure - like the gentle sensation of clothing fabric on our skin, or the firm clasp of our watch on our fist.
In order to study this, Ardem Patapoutian and coworkers decided to examine a certain variety of neurons that produced electrochemical currents (due to the flow of ions similar to the nociceptors), when force was applied on the cell surface using a glass probe. These neurons were hence sensitive to mechanical pressure. Now, assuming that these neurons too sensed pressure utilizing a membrane protein, they conducted an ingenious experiment. They blocked the expression or ‘silenced’ all the genes that were known to code for specific membrane proteins. The silencing of the right gene should render these neurons incapable of sensing pressure!
To their pleasant surprise, they identified a specific gene which when silenced, dramatically reduced the pressure perceptions in these neurons. The protein coded by this gene was thereby named Piezo, a Greek word for ‘pressure’. This protein, similar to TRPV1, was found to be localised at the cell surface and sensed pressure by allowing an influx of calcium and sodium ions. However, the most compelling evidence for Piezo’s pressure sensing abilities was revealed in experiments using mice. Mice that were designed to lack Piezo had a profound loss of touch and had severely uncoordinated body movements.
On the surface, these discoveries may seem like a drop in the ocean. However, from a research and therapeutics standpoint, they can undoubtedly have a tremendous impact.
Besides therapeutic translation, these discoveries give us exciting insights into how our body perceives the world around us. Sensation might appear to be an effortless functionality of our sense organs, however there indeed exists a whole world of proteins in our cells that orchestrate this functionality. Discoveries like this give us a humbling glimpse into this inner world of our cells.